The Ideal Gasbag Law
A colleague in passing recently noted, of a committee we both endure, that "work expands to fill the amount of space allotted", and I casually remarked that it sounded a lot like a gas.
This got me thinking.
There is, as highschool science graduates may vaguely recall, a law called the Ideal Gas Law, which is a combination of several other laws about gases that says, basically, that pressure, temperature and volume covary according to a constant for each gas.
This sounds a bit more complex than it need be, and even someone as unmathematical as I am can understand this - each value varies against the other, with a number thrown in for the number of molecules, and a constant for the gas:
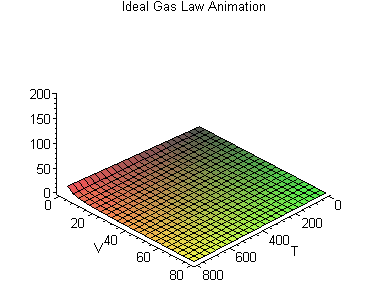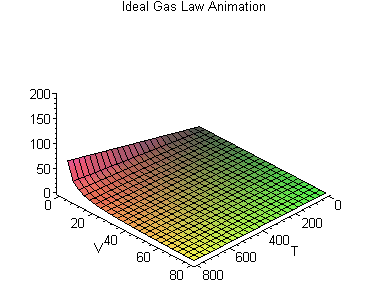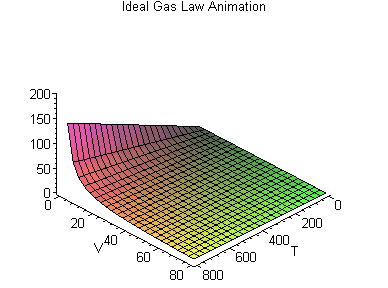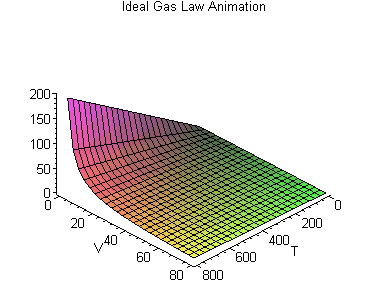
PV = nRT
or, Pressure (P) times Volume (V) equals the number of molecules in the container (n) times a constant (R) times the temperature (T).
This forms a three dimensional space that varies according to each gas constant R, in which we can say how, given two values (say, the temperature and the volume) we can work out what the third value (say, the pressure) is.
Anyway, enough real science. We are considering the science of committees. Committees have a number of "laws" attached to them, some of the best being summarised by the BBC's wonderful series from the 1980s, Yes, Minister and Yes, Prime Minister, in which the hapless politician Jim Hacker is instructed in the ways of the British Civil Service. One rule, never to be forgotten, is that whoever writes the minutes, controls what happened and what was decided.
Back to our law... a committee is, in a very real sense, a collection of particles that like gas particles under the Ideal Gas Law bounce off each other. I have been to several meetings where this was not a metaphor, but I no longer work for local government or trade unions. Perhaps the Ideal Gas Law has an analogy in the behavior of committees.
Gases expand to fill the space of their containers; so do committees. Gases become energetic when heat is applied; so do committees. Gases become agitated when the pressure is applied; and so do committees. There must be something to this.
Let us therefore see if we can make this work. Our equation is
PV = nRT.
P is pressure, that is clear. Nobody who has ever worked in a committee can fail to note the occasional application of pressure from without - and the more pressure applied, from those who rely on its achievements (poor sods), the more activity results. Not, necessarily, actual work, for just like a boiler, a committee can be harnessed or allowed to run furiously in one place, venting steam.
V, or volume in the case of gases, is the space and resources allowed for the committee to do the work it is set up to do. A good many committees are given very little room to move, or too few members (n in the equation) to do it with. You will get no effect if the number of molecules in the container is but a handful. Likewise we know that understaffed committees are unable to do much.
Some committees are staffed by ns from other containers (sorry, committees) which means that when they are in another committee doing work there, they are unavailable for work in this one. So far the analogy is holding.
We know what T is - it is the heat applied to the gas, and equally to the committee. When management or staff or regulatory bodies really do want work out of the boiler, err, committee, they use metaphors like "apply the blowtorch" or "put a fire under it". We might very well be dealing with the steam trains of the past here.
But what is R? What is the constant that determines how the committee will react? I suggest that it is the chairman (a term derived not from any gender-related language, but from the Latin term for hand, lest you think I am perpetuating the paternalistic hegemony. Well, I do that too, but only in my spare time). The ability of a chairman to constrain or direct behaviour, or in other words "stand firm" is to committeers what atomic number is to molecules.
So, Asimov's psychohistory, so long the domain of science fiction, has finally begun. We can make some predictions about how committees will behave so long as we can isolate the values of each variable. This will, I put it to you, help management, government and industry no end. And as originator of this law, which I dub the Ideal Gasbag Law, I get a 2% royalty of the profit each time it is used in the social engineering of modern society. I'll call you from the Caymans, I promise.
But there's more, as there ought to be for any good scientific explanation. First of all, there is the way the physical law fails. No gas is and ideal gas - molecules will stick together under weak forces at low temperatures and high pressures. Eventually they can become liquids (and thus uncontainable unless you have a non-porous, or legally binding, container), or even solids. We all have experience of committees which have not met or made a decision in years. The only thing to do with frozen gases like steam is put them in alcohol (preferably 20 year old scotch), and the same thing applies here, too. They may as well be used for something, even if only entertainment.
Moreover, at high temperatures, gases sublimate or even become totally different - plasma in many cases. This is a highly dangerous form of matter - it can cut through steel containers and American legal contracts, and is to be used only by experts, such as NASA or the CIA. In either case, at the extremes the gas fails to apply the law properly, something also true of the CIA. People are not uniform hard particles. They have soft squishy bits that make a mockery of simple laws. Remember that.
Also, we can use this law to do useful work - the laws of thermodynamics allow engineers (and here, corporate executives, or, in the case of democratic governments or military organisations, corporate executives) to design systems to do work. A steam engine is one such engine. So too is a committee employed efficiently, like a lobby group or a Star Chamber.
So we can now begin to take matters in hand, armed with this law, and begin to bring justice and equity to accounts departments everywhere. Particularly mine. Leave the cheque under the mat, thanks. And if anyone wants an ideal gasbag, I can name a few, in private, though I'll deny doing so in a court of law. Cheers.
© 2004, John Wilkins
